Overview
My research experience spans environmental engineering, bioinorganic chemistry, and polymer science, with a current emphasis on soft materials, polymer synthesis, and structure–property relationships.
I have 9+ years of combined academic and industry experience translating complex experimental results into clear technical reports and presentations for federal agencies, corporate sponsors, and cross‑functional scientific teams.
I’m excited to apply my understanding of structure-property relationships of soft materials and characterization to support applications development, scale-up, and technology transfer across specialty chemicals, food materials, and biomaterials. I also love creating custom tools and clear workflows that make experimentation faster, more reproducible, and easier to share across teams.
Technical Toolkit
- Polymer & Materials Characterization: GPC/SEC-MALS (molecular weight), ¹H NMR (structure/composition), DSC (Tg/Tm), TGA (thermal stability), DMA (viscoelasticity), MALDI‑MS (oligomers/mass distribution).
- Mechanical & Product‑Relevant Testing: Tensile properties (stress–strain, modulus), adhesion/hardness/abrasion, contact angle, interfacial surface tension.
- Formulation, Coatings & Thin Films: Soft‑materials formulation (gels, emulsions, dispersions), coatings & thin films (solution prep, film casting), thermal curing.
- Experimental Design, Data & Documentation: Technical data sheets; technical reports & presentations for corporate partners and federal agencies; custom research tools and well‑documented workflows for reproducibility.
- Collaboration, Leadership & Training: Consulting and training for startup leadership (including CTO), mentoring undergraduates and graduate students, cross‑functional communication across R&D, applications, and external partners.
Presentations
Alternative Solvents for Metal-Free Ring-Opening Metathesis Polymerization
Ring-opening metathesis polymerization (ROMP) is a type of polymerization that is industrially relevant. Typically, ROMP uses expensive transition metal catalysts for the polymerization. This can be problematic for applications that require little to no metal contamination.
One method to eliminate metal species in polymers is metal-free ring-opening metathesis polymerization (MF-ROMP). MF-ROMP is a type of polymerization that uses all organic reagents and can be activated/deactivated by blue light. MF-ROMP typically uses dichloromethane (DCM) as the solvent, but there has been increased interest in reducing DCM in industrial chemical processes due to environmental and safety concerns.
We identified alternative solvent systems for MF-ROMP and probed the properties that make a solvent suitable for MF-ROMP. We demonstrate the effects of different solvent systems on MF-ROMP for a few industrially relevant ROMP monomers. This work will help identify a more environmentally friendly way to make polymers by MF-ROMP.
Presented at 2025 National Graduate Research Polymer Conference at Arizona State University as part of the “Green chemistry approaches to polymer design and synthesis” session

Controlling Crosslinking density of Linear Metal-Free Ring-Opening Metathesis Polymers
Crosslinking density impacts the material properties of thermosets, but spontaneous crosslinking between alkenes and reactive metal species in metal-mediated ROMP makes it difficult to control the crosslinking density.
Polydicyclopentadiene (PDCPD) is a common thermoset comprised of a crosslinked network of polymer chains. Although the crosslinked network makes PDCPD a high impact resistance material, the inability to control the crosslinking limits the range of material properties.
We investigated how crosslinking density can be controlled by studying the effects of the composition and weight percent loading of linear PDCPD copolymers on the crosslinking density and material properties, such as Tg and mechanical properties. This work gives insights on how to improve ductility and temperature stability of high-energy dissipating materials for ballistic performance.
Presented at American Chemical Society Fall 2023 San Francisco SciMix Poster Session
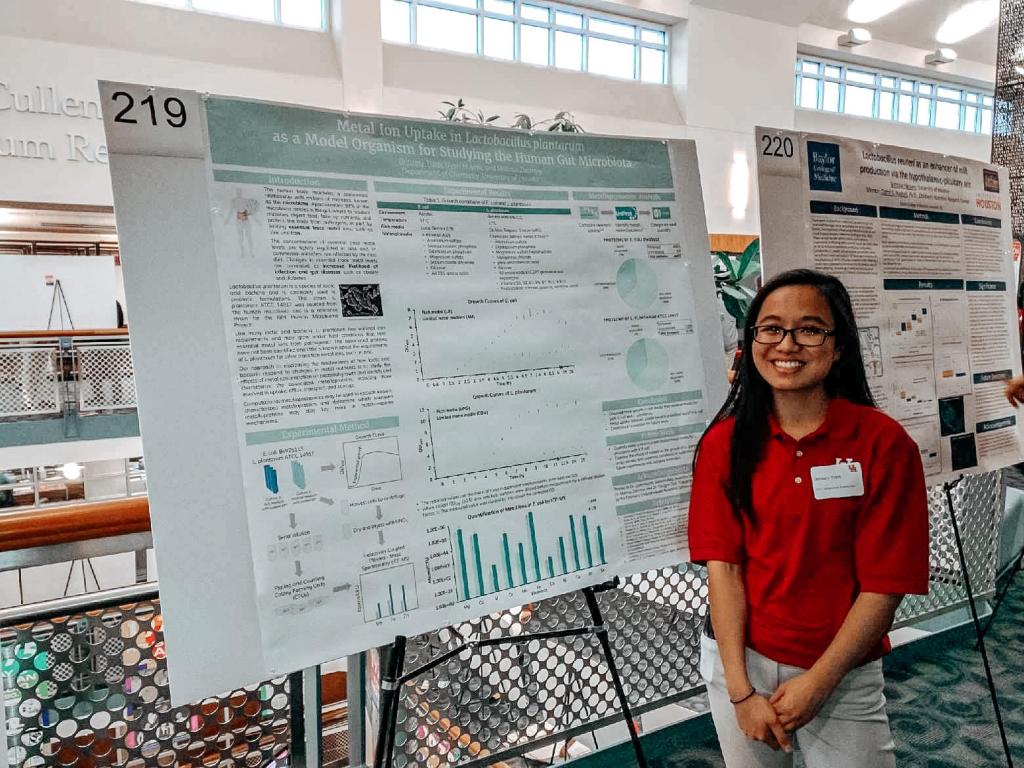
Metal Ion Uptake in Lactobacillus Plantarum as a Model Organism for Studying the Human Gut Microbiota
The human body maintains a commensal relationship with millions of microbes, known as the microbiota. Most of these microbes are found in the gastrointestinal tract, where they digest food, take up nutrients, and protect the human body from pathogens. Changes in essential trace metal levels, including manganese, zinc, and iron, influence the abundance of different types of microbial species and strongly correlate to altered infectious disease susceptibility.
Despite recent efforts to map microbiota chemical functions, the role of metals in this area remains underdeveloped. To determine how different gut bacteria can tolerate changes in essential trace elements, we developed gut bacteria culture methods for growth and quantified the metal ion uptake of E. coli. This work will help improve current therapies for gut diseases and potential targets for development of new therapeutics.
Presented at University of Houston 2018 Undergraduate Research Day
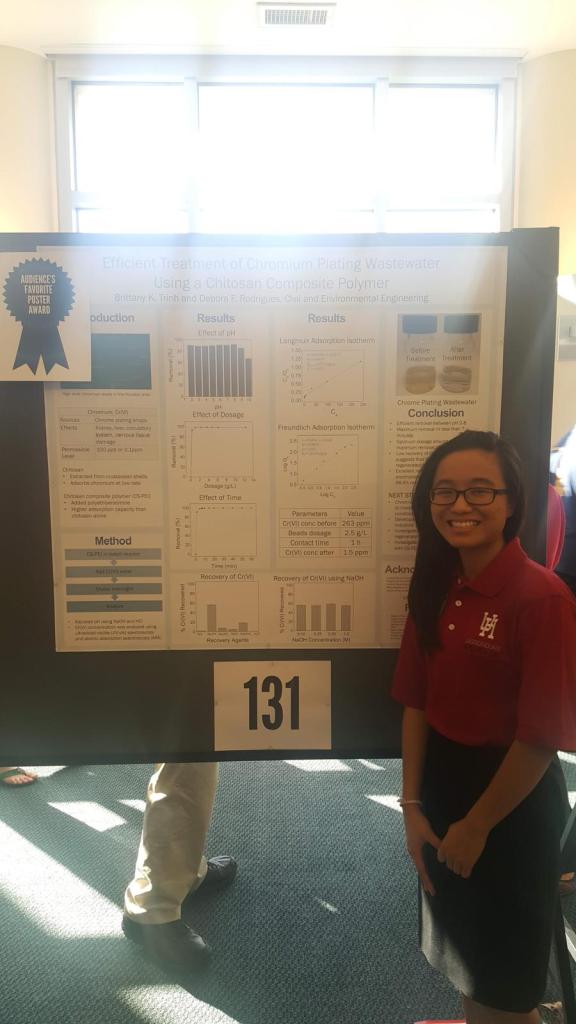
Efficient Treatment of Chromium Plating Wastewater Using a Chitosan Composite Polymer
Hexavalent chromium (Cr (VI) is a highly toxic form of chromium, primarily produced by industrial processes, like chrome plating. Cr(VI) can affect health by damaging the kidney, liver, circulatory system, and nervous tissue damage. Polymers can be used as adsorbents for direct or indirect treatment of wastewater.
In this work, we incorporate chitosan (CS), which can be extracted from crustacean shells, with polyethyleneimine (PEI), to make a polymer composite (CS-PEI). We successfully used 2.5 g/L of CS-PEI to reduce Cr(VI) content from 263 ppm to 1.5 ppm within 1 hour. Additionally, the CS-PEI could be recovered and reused. This work provides a method to remove heavy metal contaminants from wastewater efficiently and effectively.
Presented at University of Houston 2017 Undergraduate Research Day

You must be logged in to post a comment.